|
11/25/2023 0 Comments Kcl lattice energy 
While in the case of NaF and KCl, NaF has a greater lattice energy compared to KCl because the size of the cation and anion is smaller in the case of KCl. What is lattice energy Learn the definition of lattice energy, see its trends in the Periodic Table, and study how to find lattice energy using its formula. Bonding & Molecular Structure Lattice Energy. The lattice energies of KF, KCl, KBr and KI follow the order : Lattice Energy q C + q A r C + + r A The incorrect order of lattice energy is In. 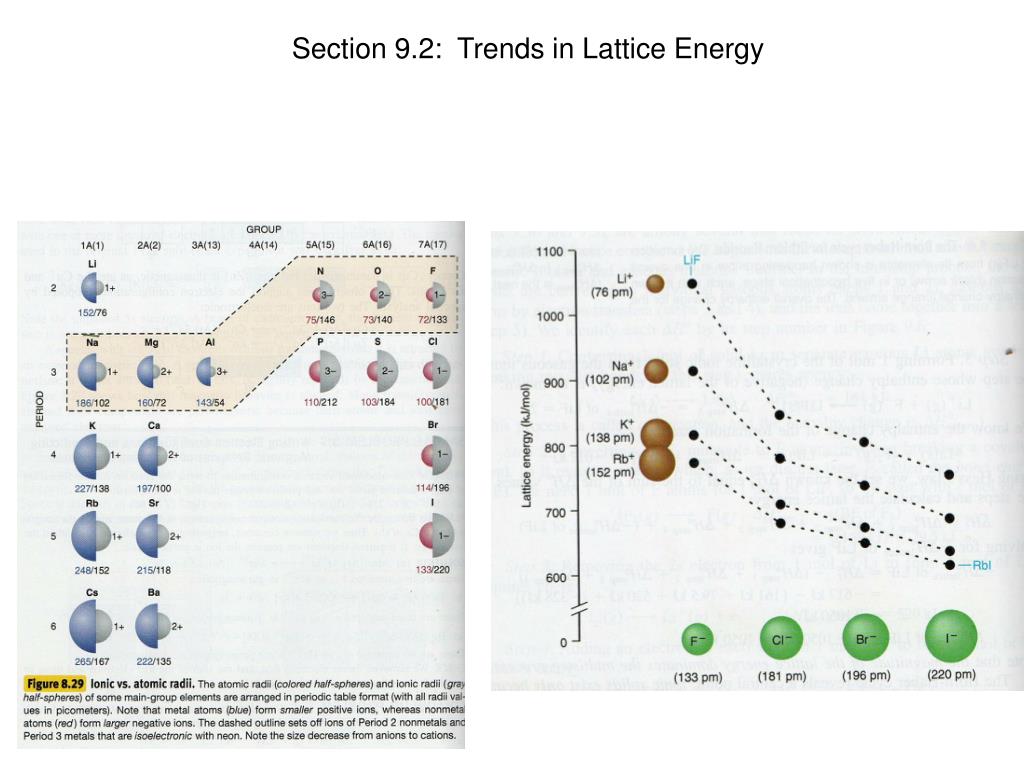
That immediately removes any possibility of confusion. Among the given substances, MgO has the highest lattice energy compared to CaO as lattice energy is directly related to the charge and size of the cation. Arrange these compounds in order of increasing magnitude of latti. For NaCl, the lattice formation enthalpy is -787 kJ mol-1. The dissolution of KCl decreases the solution temperature which suggests that it is an endothermic reaction, and the energy of dissolution will be positive. Explain why the lattice energy for sodium chloride is less (i.e., more negativ. You should talk about 'lattice formation enthalpy' if you want to talk about the amount of energy released when a lattice is formed from its scattered gaseous ions. The lattice energies (Delta Ho lattice) for sodium chloride and potassium chloride are -788 kJ and -717 kJ, respectively. higher ionization of lithium is more than compensated by the higher lattice energy and pushing the equilibrium as potassium vapor partial pressure is being decreased by vapor condensation. For NaCl, the lattice dissociation enthalpy is +787 kJ mol-1. Which has highest lattice energy Because the ionic radii of the cations decrease in the order K + > Na + > Li + for a given halide ion, the lattice energy decreases smoothly from Li + to K +. Thermodynamic parameters are shown in the table below. KCl will have the more negative lattice energy because potassium is smaller than cesium and they both have a 1 + charge. With potassium vapors being eliminated by condensation on colder surfaces of the apparatus, used in the video. That is because higher ionization energy is more than compensated by more negative hydration enthalpy of $\ce$$ As Ca, S have 2 units of charge while K, Cl have one and there is not much of a difference in size ( atleast not enough to overcome the doubling of the charge), the charge density of Ca and S ions would be more. But lithium has more negative standard redox potential (which applies in water context only). The Lattice Energy of Ionic Compounds is directly proportion to the Charge Density. Place the following in order of decreasing magnitude of lattice energy: a. Here are the lattice energies for NaCl, KCl, and MgCl2. Question: Place the following in order of decreasing magnitude of lattice energy: a) K2O, Rb2S, Li2O b) NaF, RbBr, KCl c) KF, CaS,RbI. Lattice energy is the energy needed to separate 1 mol of a solid ionic compound into gaseous ions. Potassium reacts more violently with water than lithium. Question: Place the following in order of decreasing magnitude of lattice energy: a) K2O, Rb2S, Li2O b) NaF, RbBr, KCl c) KF, CaS,RbI.  
TNT is much more reactive than coal, but released specific energy during coal burning is 10 times bigger than during TNT explosion. Reactivity (kinetics) and tendency of reaction outcome (thermodynamics) are independent. \( \newcommand\)).Note that element electronegativity $\chi$ is a combined indicative quantity from covalent bond context, so its comparative values do not always apply in scenario analysis what happens or should be preferred. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
